MEPSEVII® (vestronidase alfa-vjbk) demonstrated improvement or stabilization across multiple measures1
Mobility
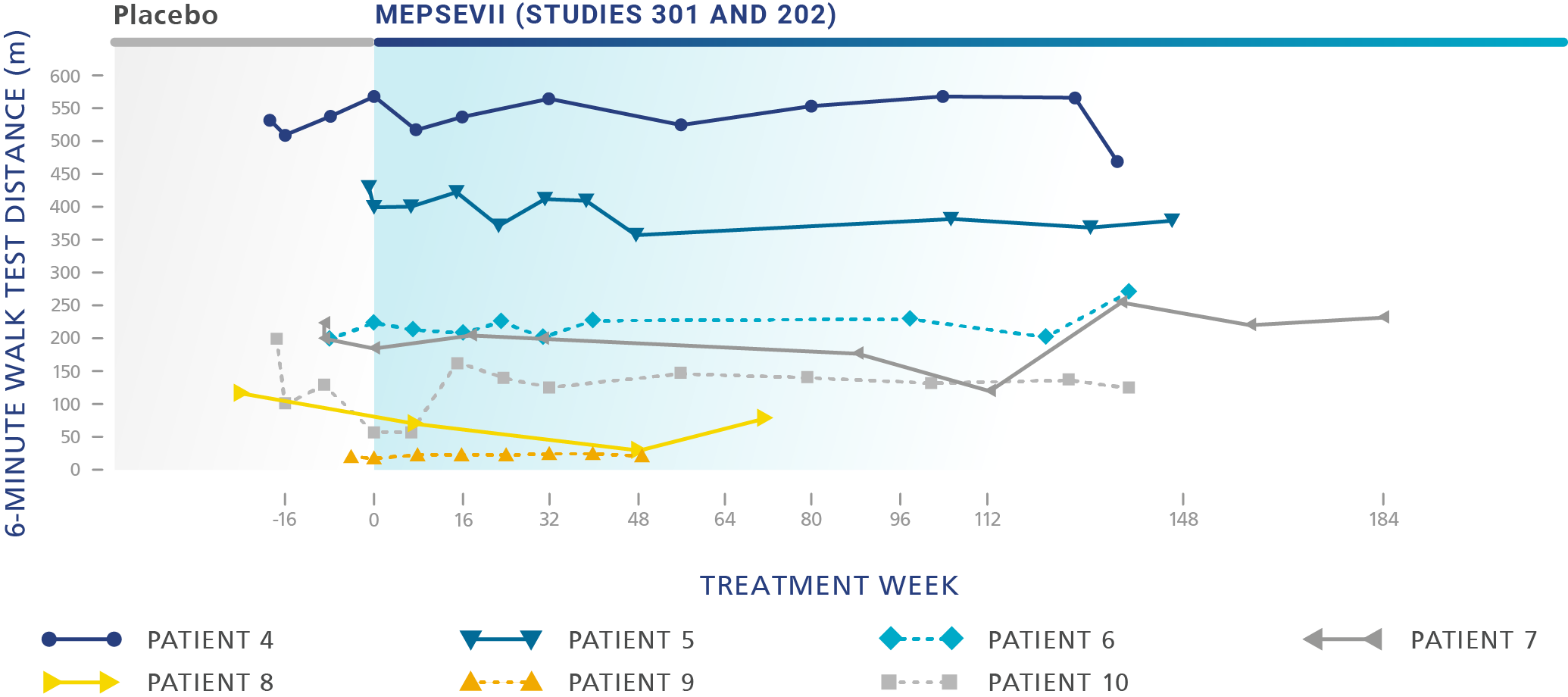
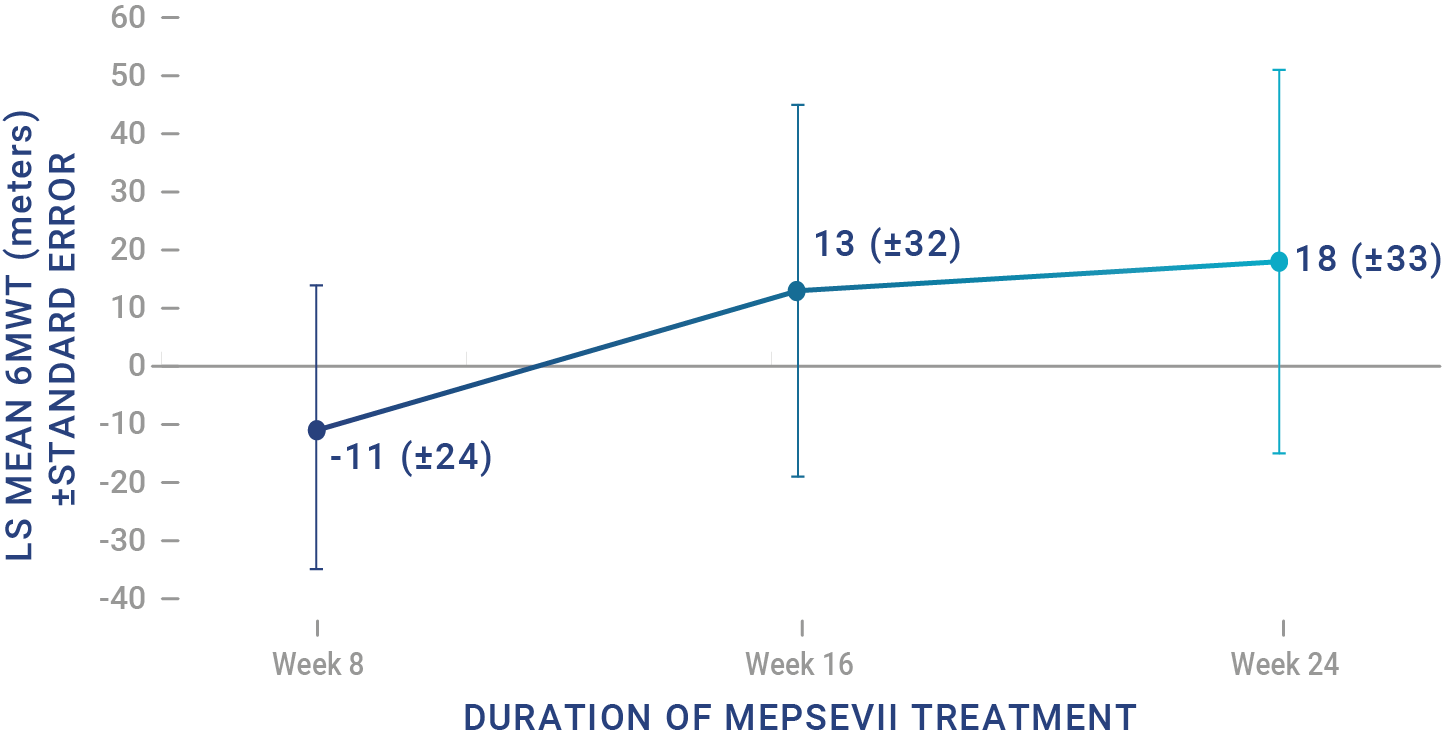
Patients treated with MEPSEVII had improved or stabilized mobility1
Respiratory
MEPSEVII demonstrated improvement in pulmonary function after 120 weeks of treatment1
Improvement in pulmonary function from baseline
21% improvement in FVC (% predicted) in 1 patient1
Study 201 - Ex-US investigation (120 weeks): A single-arm, open-label, dose-exploration study outside the United States included 3 patients with MPS VII aged 5 to 25 years. FVC = forced vital capacity.
Mobility in this patient also improved1
- 105-meter improvement in the 6MWT in the same patient
Small heterogeneous patient group1
- Due to the extremely small population of patients with MPS VII, the patient group was heterogeneous
- Clinical end points in some patients were not assessable due to the extent of disease, age, or level of cognition
Study 301 - Randomized Start trial (Baseline to 48 weeks): The efficacy study was a randomized-start, phase 3 clinical study in which 12 patients (4 males, 8 females) aged 8 to 25 years (median age: 14 years) were assessed to evaluate the efficacy and safety of recombinant human ß-glucuronidase (rhGUS) enzyme replacement therapy (ERT) in patients with mucopolysaccharidosis VII (MPS VII).1,2
Study 203 - Open-label, single arm (48 weeks): An open-label, uncontrolled single arm study in which 8 patients below 5 years of age received MEPSEVII at 4 mg/kg every 2 weeks for 48 weeks with an optional 240 weeks of additional dose continuation period.1
Study 201 - Ex-US investigation (120 weeks): A single-arm, open-label, dose-exploration study outside the United States included 3 patients with MPS VII aged 5 to 25 years.1
Small heterogeneous patient group1
- Due to the extremely small population of patients with MPS VII, the patient group was heterogeneous
INDICATION
MEPSEVII is a recombinant human lysosomal beta glucuronidase indicated in pediatric
and adult patients for the treatment of Mucopolysaccharidosis VII
(MPS VII, Sly syndrome).
Limitations of Use
The effect of MEPSEVII on the central nervous system
manifestations of MPS VII has not been determined.
BOXED WARNING AND ADDITIONAL IMPORTANT SAFETY INFORMATION
WARNING: ANAPHYLAXIS
- Anaphylaxis has occurred with MEPSEVII administration, as early as the first dose, therefore appropriate medical support should be readily available when MEPSEVII is administered.
- Closely observe patients during and for 60 minutes after MEPSEVII infusion.
- Immediately discontinue the MEPSEVII infusion if the patient experiences anaphylaxis.
- Anaphylaxis to MEPSEVII was reported in 2 of 20 patients in the clinical program. The two patients with anaphylaxis to MEPSEVII during the clinical trials had one occurrence each and tolerated subsequent infusions of MEPSEVII, without recurrence.
- Consider the risks and benefits of re-administering MEPSEVII following anaphylaxis
- Manifestations included respiratory distress, cyanosis, decreased oxygen saturation, and hypotension.
- Prior to discharge, inform patients of the signs and symptoms of anaphylaxis and instruct them to seek immediate medical care if symptoms occur.
Adverse Reactions
- In a clinical trial, the most common adverse reactions occurring with MEPSEVII treatment included infusion site extravasation, diarrhea, rash, anaphylaxis, infusion site swelling, peripheral swelling, and pruritus.
- One patient experienced a febrile convulsion during MEPSEVII treatment. The patient subsequently was re-challenged without recurrence and continued on treatment.
Use in Specific Populations
- There are no available data on MEPSEVII use in pregnant women to determine a drug-associated risk of adverse developmental outcomes.
- There are no data on the presence of MEPSEVII in either human or animal milk, the effects on the breastfed infant, or the effects on milk production.
You may report side effects to the FDA at (800) FDA-1088 or www.fda.gov/medwatch.
You may also report side effects to Ultragenyx at 1-888-756-8657.
Please see full Prescribing
Information, including the BOXED WARNING,
for a complete discussion of the risks associated with MEPSEVII.
References: 1. MEPSEVII [package insert]. Novato, CA: Ultragenyx Pharmaceutical Inc; 2020. 2. Data on file. Clinical Study Report UX003-CL301. Ultragenyx Pharmaceutical Inc; January 9, 2017.

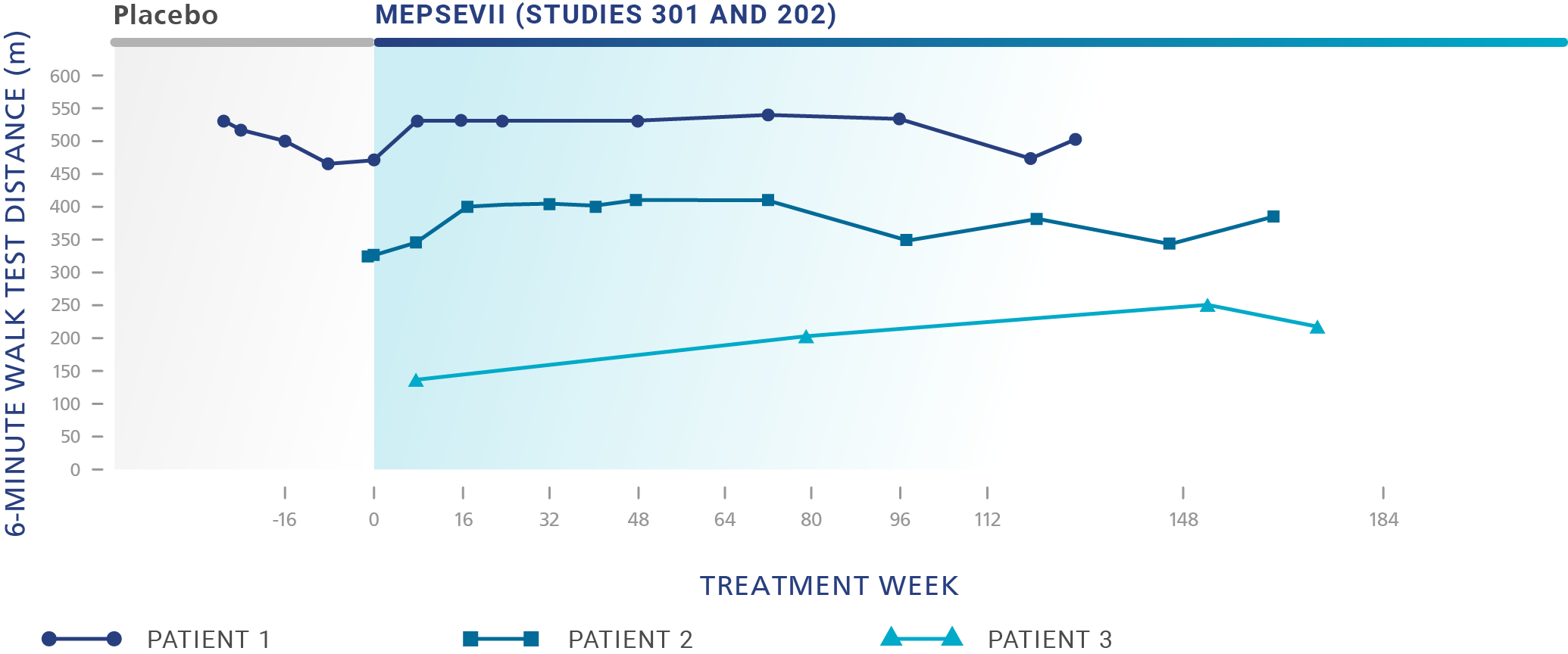
 Study 301 ‑ Randomized Start trial (Baseline to 48 weeks): The efficacy study was a randomized-start, phase 3 clinical study in which 12 patients (4 males, 8 females) aged 8 to 25 years (median age: 14 years) with MPS VII were assessed to evaluate the efficacy and safety of recombinant human β-glucuronidase (rhGUS) enzyme replacement therapy (ERT).1,2
Study 301 ‑ Randomized Start trial (Baseline to 48 weeks): The efficacy study was a randomized-start, phase 3 clinical study in which 12 patients (4 males, 8 females) aged 8 to 25 years (median age: 14 years) with MPS VII were assessed to evaluate the efficacy and safety of recombinant human β-glucuronidase (rhGUS) enzyme replacement therapy (ERT).1,2